Abstract
Decitabine (Dec) and Azacitidine (Aza) that target DNA methyltransferase 1 (DNMT1) are hypomethylating agents (HMAs) approved to treat acute myeloid leukemia (AML) in combination with Venetoclax (Ven). The combination is also used to treat high-risk myelodysplastic syndromes, especially TP53-mutated (TP53mut) cases in which responses to HMA alone are short-lived. In most patients (pts), however, myelosuppression from treatment leads to frequent Ven duration and/or dose-reductions, and/or cycle delays. An approach to decrease HMA-mediated myelosuppression but maintain S-phase dependent DNMT1-targeting, evaluated in a previous clinical trial (https://doi.org/10.1111/bjh.16281), is to administer noncytotoxic doses/concentrations of Dec (0.2 mg/kg; ~5 mg/m 2) by a frequent-distributed schedule of 1X/week. An approach to decrease Ven mediated myelosuppression but maintain cooperation with HMA, shown in pre-clinical studies, is to administer a single-dose prior to HMA. Ven can depolarize mitochondrial membranes; mitochondrial membrane-potential is essential to function of the mitochondrial enzyme DHODH that produces cytidine/deoxycytidine that competes with HMA in cells. Thus, Ven prior to HMA dosing temporarily inhibits de novo pyrimidine synthesis, to counter a major mechanism of resistance to HMA in MDS/AML, without suppressing normal myelopoiesis (https://doi.org/10.1182/blood-2020-143200). We conducted a retrospective analysis of all pts with TP53mut MDS or AML treated with weekly Ven and low-dose subcutaneous Dec at our institution. We analyzed the characteristics of these pts, response to therapy, and outcomes using standard descriptive statistics. Mutational testing was performed using a commercial next-generation sequencing (NGS) panel.
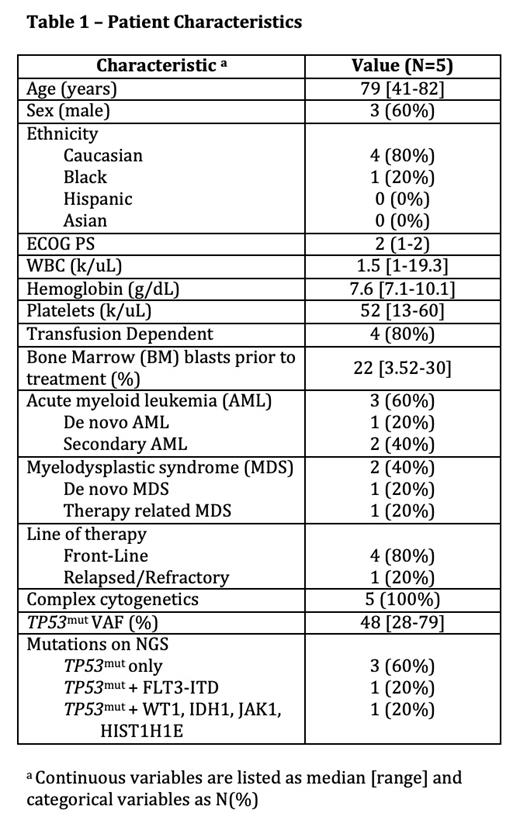
Five pts, 3 male and 2 female, with TP53mut MDS or AML were treated with weekly Ven 400 mg on D1 and subcutaneous Dec 0.2 mg/kg on D2, administered weekly in 28 day cycles. Two pts had MDS (1 de novo, 1 treatment related) and 3 pts had AML (1 de novo, 2 secondary from prior MDS). Four pts (80%) received the treatment in frontline, all with poor performance status (PS), and 1 pt (20%) had R/R disease. Median age at diagnosis was 79 years [41-82]. The only young pt had prolonged severe cytopenias after 1 cycle Dec standard dosing during the peak of COVID-19 pandemic so was switched to this regimen. Of the 4 frontline treated pts, 2 pts had high-risk MDS, and 2 pts had adverse risk AML. The R/R pt had high-risk MDS transformed to AML that was refractory to 2 prior lines of therapy: standard Aza/Ven x5 cycles, then standard Vyxeos. Disease cytogenetics were complex in all pts. 60% (3/5) pts had sole TP53mut on NGS, with median variant allelic frequency (VAF) 48% [28-79]. 80% (4/5) pts were transfusion dependent prior to treatment. Median time to initiating therapy was 7 days from initial or refractory diagnosis [3-59] and median follow-up was 7.8 months (mo) [2.9-11.4]. The overall response rate (ORR) was 100%: 4/4 frontline pts had complete remissions (CR), and the 1 R/R pt achieved morphologic leukemia-free state (MLFS). Median time to best response was 2.9 mo. 50% (2/4) pts became transfusion independent. 40% (2/5) pts lost their TP53mut at best response, and another 40% (2/5) pts had significant reductions (83% and 38%) in TP53 mut VAF. The regimen was well tolerated with no pts stopping therapy due to adverse effects (AE) . AE included G3/G4 neutropenia (80%), G1 thrombocytopenia (40%), nausea (20%), fatigue (20%), lower extremity edema (20%), pneumonia (60%), and neutropenic fever (20%) with a median of 1 unplanned hospitalization per pt during follow-up. 60% (3/5) pts remain in CR on continued therapy for a median of 7.8 mo [7.2-9.4] thus far. One pt underwent allogeneic stem cell transplantation, however, died 11.4 mo after conditioning due to transplant related mortality. The R/R pt died after being lost to follow-up 2.9 mo after therapy initiation. No pt had measurable relapse during follow-up.
Combination weekly Ven with subcutaneous low-dose Dec is well tolerated yielding high rates of clinical and molecular response in pts with TP53mut MDS/AML. Although small, this case-series extends previous clinical trial proof-of-activity of non-cytotoxic DNMT1-targeting to a high-risk, poor PS, historically chemorefractory patient population. The regimen allowed frequent, sustained exposure to therapy often not possible with standard HMA/Ven regimens.
Shastri: Kymera Therapeutics: Research Funding; Guidepoint: Consultancy; GLC: Consultancy; Onclive: Honoraria. Gritsman: iOnctura: Research Funding. Feldman: Glycomimetics: Current Employment, Current holder of stock options in a privately-held company. Verma: Celgene: Consultancy; Acceleron: Consultancy; Novartis: Consultancy; Stelexis: Consultancy, Current equity holder in publicly-traded company; Eli Lilly: Research Funding; Curis: Research Funding; Medpacto: Research Funding; Incyte: Research Funding; GSK: Research Funding; BMS: Research Funding; Stelexis: Current equity holder in publicly-traded company; Throws Exception: Current equity holder in publicly-traded company. Saunthararajah: EpiDestiny: Consultancy, Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal